We are happy to announce that we have renewed our ISO 13485:2016 certification for 3 years. This is the result of the hard work and daily commitment of our teams.

LATEST NEWS
We are expanding our range of services and industrial capabilities with the acquisition of the French company Medical Tubing.
Based in Le Bousquet d’Orb, in the Hérault region, Medical Tubing offers its customers 30 years of experience in thermoplastic extrusion. The company serves all medical markets with tubes of all sizes, multi-layer and multi-lumen. It also offers compounding of multiple resins for greater responsiveness. With Medical Tubing, we complete our offering as a solutions integrator for all single-use medical device and biopharmaceutical development projects. Following the acquisition in 2022 of A. Hopf GmbH, a German company based in Cadolzburg, we now cover the entire value chain for single-use life science devices, with more than 3,300 sqm of ISO 8 cleanroom space : These investments in France and Europe in the medical sector enable us to offer European and global medical device players a single point of entry for their projects, reducing development times and reducing costs and integrating the challenges of industrialisation and marketing from the outset.
We are proud to announce the acquisition of A. Hopf Kunststoffverarbeitung GmbH, a family-owned German company based in Cadolzburg, specialising in plastic injection moulding for the life sciences sector.
Founded in 1949 by Mr Anton Hopf, A. Hopf GmbH was one of the pioneers of injection moulding in Germany. Since then, the family name has been associated with high quality and customised solutions for single-use life science components. With a 1600 sqm production area, 350 sqm of ISO 8 clean room and a 350 sqm tool shop, A. Hopf GmbH serves the whole of Europe from its base in Germany. The company offers a range of components for enteral and intravenous applications and any medtech activity (components for nutrition, high flow valves, production of specific parts in thermoplastics, precision injection moulded parts). Any thermoplastic material can be processed in Cadolzburg, with a focus on BPA-free solutions. In this way, A. Hopf enables us to integrate the injection moulded parts into your overall projects and respond to your needs.


Thanks to our unique manufacturing expertise, our R&D team has helped our client, a contract manufacturer, to review and improve a critical production process manufacturing equipment.
DEVELOPMENT PROCESS → PROJECT Manufacturing Process Analysis
→ OUTCOME Design History File
This has resulted in improved capabilities and quality of products delivered.
Mechanical CAD Design
Industrial Automatism Software Development
Equipment Manufacturing
FMEA Risk Analysis
IQ OQ PQ Validation Plan
CE Mark
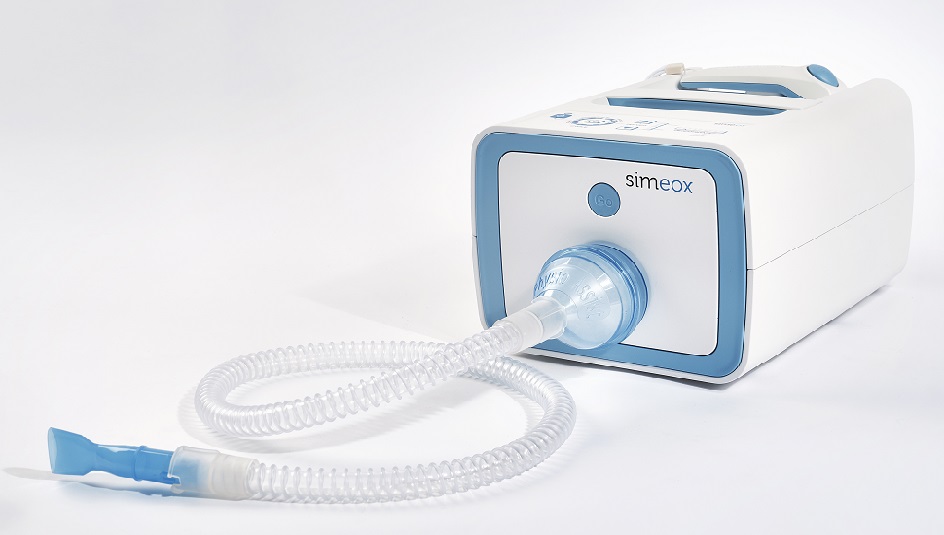
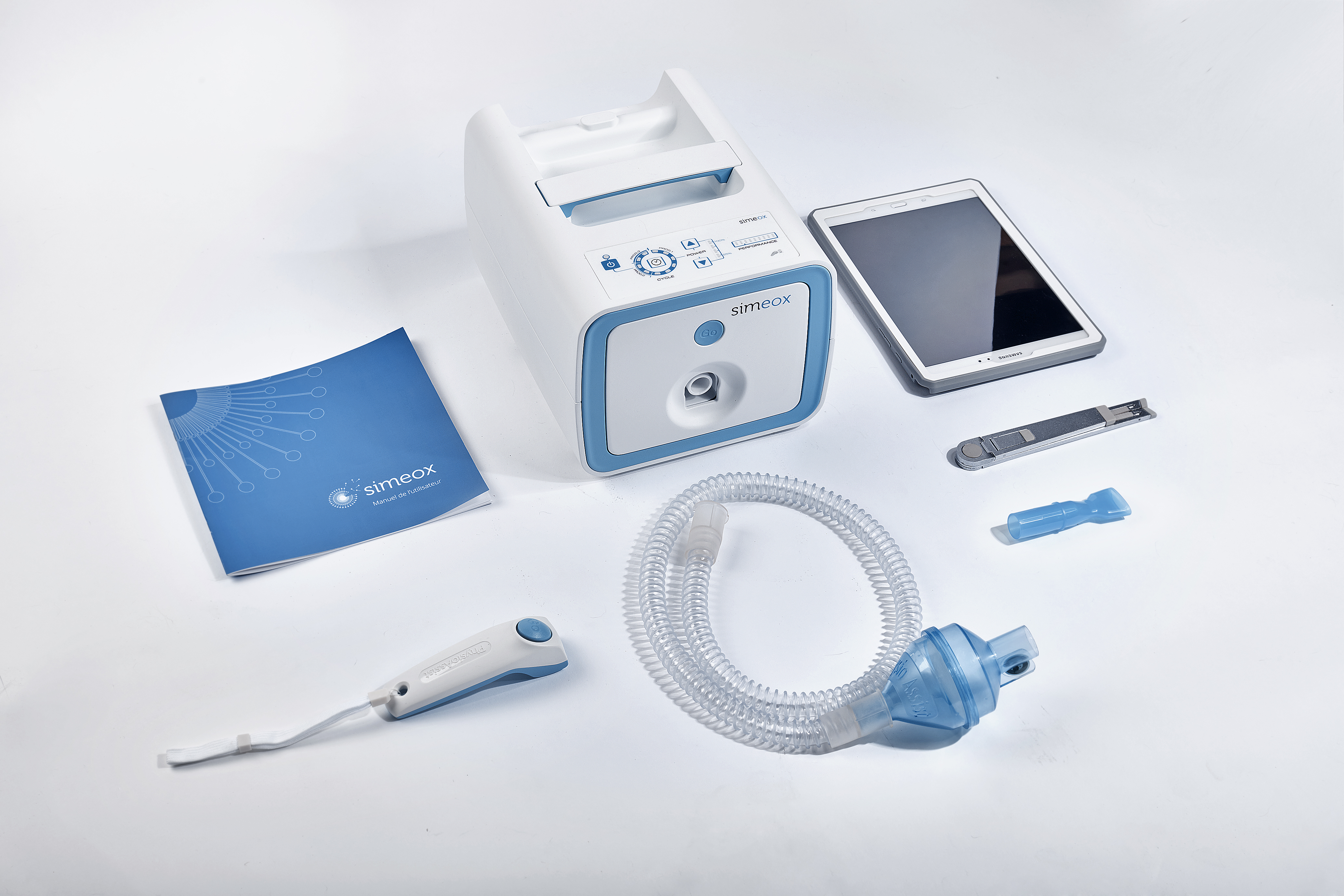
Our client, Physio-Assist has developed an innovative concept of equipment and tubing set dedicated to bronchial pathologies. We have acted as their product development and industrialization partner and have help drive the concept to fully operational equipment.
DEVELOPMENT PROCESS → EQUIPMENT PROJECT Design for Manufacture
→ DISPOSABLE DEVICE PROJECT Design for Manufacture
→ OUTCOME Design History File
→ MANUFACTURING Injection Molding
Physio-Assist:
A French innovative company search a partner for equipment and disposable device development.
Mold Manufacturing
FMEA Risk Analysis
Material Selection
Mold and Assembling Tools Manufacturing
FMEA Risk Analysis
IQ OQ PQ Validation Plan
Performance Validation
CE Mark – OBL Contract
Ultrasonic Welding
Assembling; Packaging
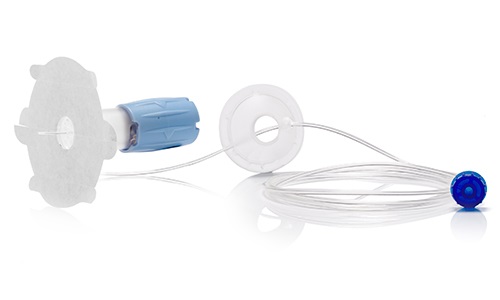
After industrialization of an initial version of the device, our R&D team has fully redesigned the new version based on competitive review of functionalities. In addition, we have co-developed and co-patented part of the product.
DEVELOPMENT PROCESS → PROJECT Concept Pre-study
→ OUTCOME Design History File
→ MANUFACTURING Injection Molding
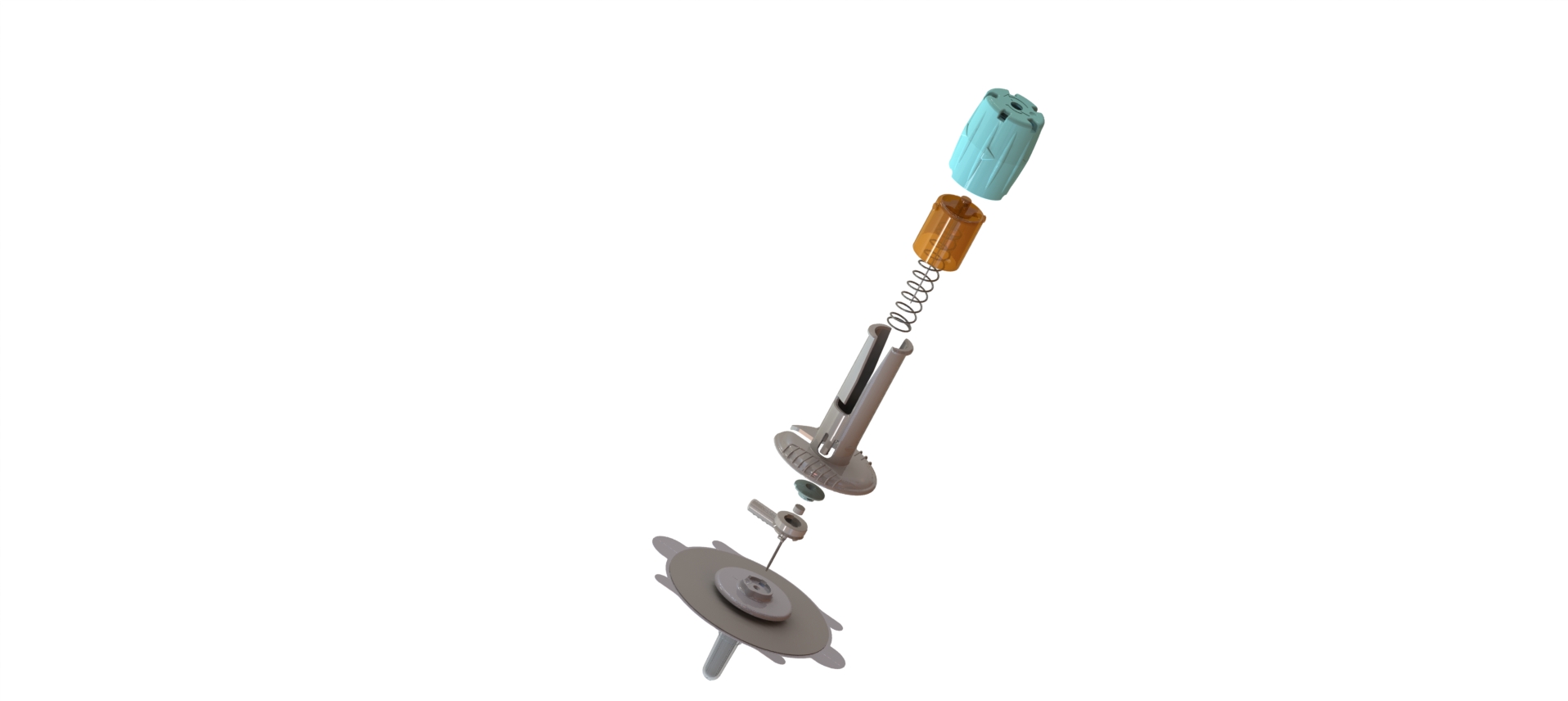
CAD Design
Rapid Prototyping
Material Selection
Design for Manufacture
Mold Manufacturing
Assembling Tools Manufacturing
FMEA Risk Analysis
IQ OQ PQ Validation Plan
CE Mark
FEP Tip Forming
Assembling
Packaging
Sterilization
For our innovative client, we have reviewed and analyzed decompression devices on the market and redefined the market standard with new design and improved performance for end users.
DEVELOPMENT PROCESS → PROJECT CAD Design
→ OUTCOME Design History File
→ MANUFACTURING Injection Molding
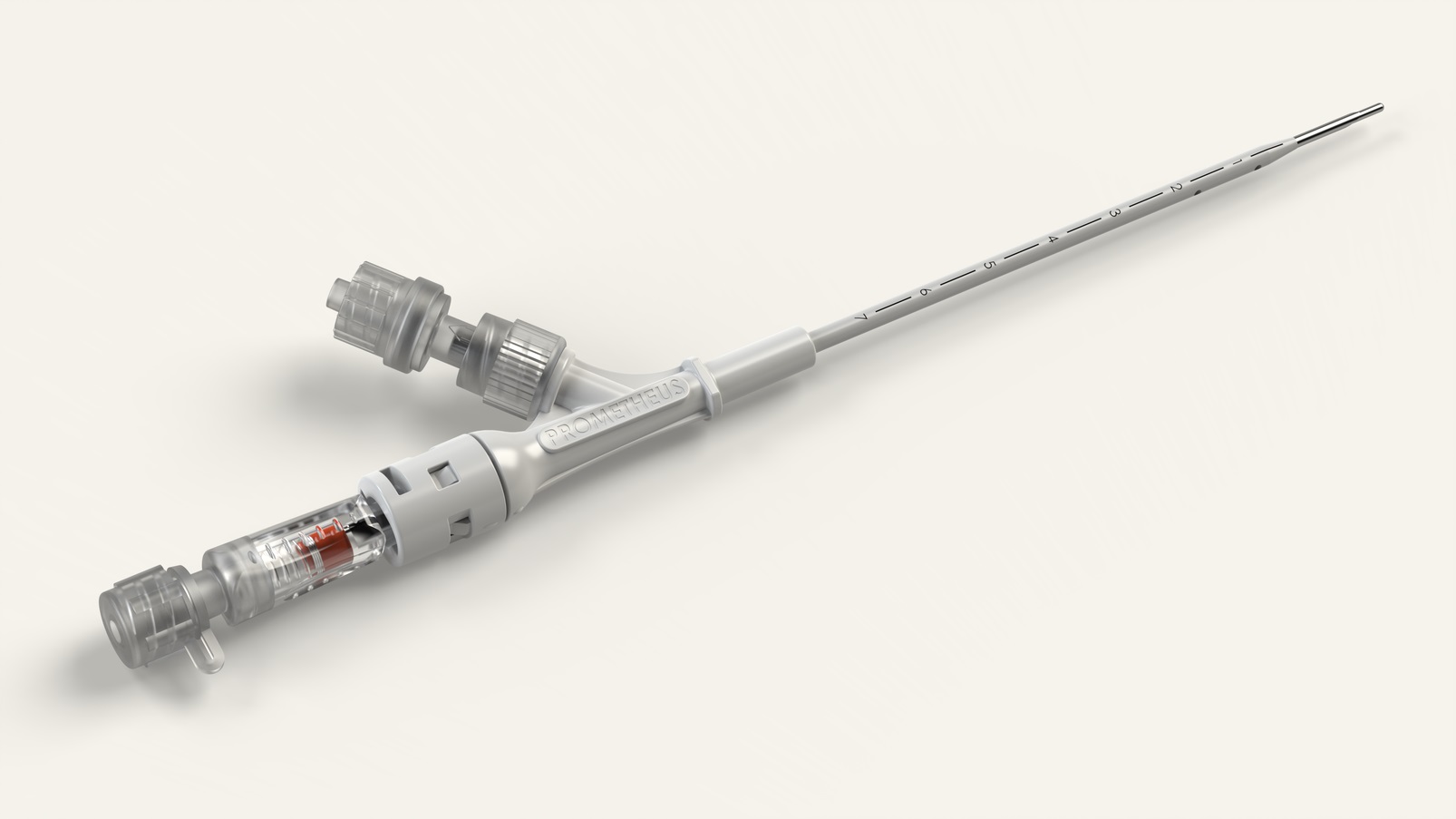
Design for Manufacture
Mold Manufacturing
Assembling Tools Manufacturing
FMEA Risk Analysis
IQ OQ PQ Validation Plan
CE Mark
Over-molding
Tip forming
Driling
Pad Printing
Assembling; Packaging; Sterilization
After the initial basic design phase, our R&D team has fully reviewed the initial prototype and help redevelop and industrialize this device to meet market requirements.
DEVELOPMENT PROCESS → PROJECT Concept Pre-study
→ OUTCOME Design History File
→ MANUFACTURING Injection Molding

CAD Design
Rapid Prototyping
Material Selection
Design for Manufacture
Mold Manufacturing
Assembling Tools Manufacturing
FMEA Risk Analysis
IQ OQ PQ Validation Plan
Performance Validation
CE Mark – OBL Contract
Ultrasonic Welding
100% Performance testing
Assembling
Packaging
Sterilization









